At the heart of Earth’s intricate interdependent physical systems lies the atmosphere, an extraordinary ensemble of gases that plays a crucial role in shaping our planet’s dynamics. This vast gaseous envelope envelops not only our home but also other celestial bodies, giving rise to a fascinating realm of atmospheric science.
The very concept of an atmosphere evokes a complex assembly of gases that encircle planets and celestial bodies. For Earth, this atmospheric cloak is predominantly a mixture of nitrogen, constituting an impressive 78%, and oxygen, which accounts for approximately 21%. In addition to these predominant gases, a myriad of other gases collectively make up the remaining 1%, contributing to the intricate balance that sustains life as we know it.
The atmosphere doesn’t conform to a uniform composition; rather, it unfolds in layers that each possess distinct characteristics. These layers—troposphere, stratosphere, mesosphere, thermosphere, and exosphere—unfold in a vertical symphony defined by varying temperature and pressure gradients. This stratified arrangement not only showcases the atmosphere’s complexity but also shapes its myriad functions.
One of its primary roles is safeguarding life on Earth. Acting as a cosmic shield, the atmosphere intercepts and absorbs the potentially harmful ultraviolet (UV) radiation emanating from the sun, preventing it from reaching the surface in excessive amounts. This protective function is not just vital for our well-being but also for the stability of ecosystems and the preservation of genetic information.
Beyond its protective role, the atmosphere actively participates in Earth’s temperature regulation. Through a process akin to a cozy blanket, the atmosphere blankets the planet, trapping heat that’s radiated from the surface. This natural insulation mechanism prevents temperature extremes, ensuring that our world remains conducive to diverse forms of life.
The interaction of sunlight with the atmosphere initiates a mesmerizing dance of convection. As the sun warms different atmospheric layers, hot air rises, cooler air descends, and a cyclic movement ensues. This convection-driven air movement is a cornerstone of weather patterns, dictating winds, storms, and climate variations. From gentle breezes that rustle leaves to mighty hurricanes that reshape landscapes, the atmosphere’s influence on our weather is profound and far-reaching.
In essence, the atmosphere is a dynamic orchestra of gases, orchestrating a symphony of life-sustaining processes. Its composition, layered structure, and intricate functions underscore its indispensable role in preserving the delicate equilibrium of our planet. As we gaze at the sky or feel the gentle caress of the wind, we are reminded of the profound interconnectedness that links us to this ethereal realm, shaping the very fabric of our existence.
Planetary Atmospheres
The diversity of our solar system extends beyond the planets themselves, encompassing their atmospheres as well. Each celestial body, from planets to a handful of moons, possesses its own atmospheric composition and behavior. Intriguingly, even exoplanets have been scrutinized through light spectroscopy to unveil their atmospheric secrets.
Within our solar system, every planet boasts an atmosphere uniquely tailored to its nature. Mercury’s atmosphere, for instance, is incredibly sparse, resembling the vacuum of space more than a conventional atmosphere. In contrast, gas giants like Jupiter, Saturn, Uranus, and Neptune showcase voluminous, profound atmospheres characterized by their thickness and depth.
On the other hand, rocky planets such as Earth, Venus, and Mars possess comparatively thinner atmospheres enveloping their solid surfaces. Moons within our system typically bear slender atmospheres, except for Titan, a moon of Saturn, where the air pressure at its surface surpasses that of Earth.
The roster of officially acknowledged dwarf planets also adds to this assortment. Pluto exhibits a fluctuating atmosphere, expanding and contracting with changing seasons. Meanwhile, Ceres, another dwarf planet, hosts an extremely delicate and transient atmosphere, predominantly composed of water vapor.
Amidst this atmospheric array, Earth stands apart with its intricate layered structure. This unique arrangement adeptly captures just the right amount of the Sun’s energy to provide warmth while simultaneously shielding the planet from detrimental solar radiation. This delicate equilibrium sustains life on our remarkable planet.
Definition of Atmosphere
The Earth is enveloped by a blend of gases known as the atmosphere. This mixture serves essential purposes: supplying breathable air, safeguarding against harmful UV radiation from the Sun, retaining heat to maintain planetary warmth, and mitigating extreme day-to-night temperature variations. While every planet in our solar system possesses an atmosphere, none mirror Earth’s atmospheric composition or layered configuration.
Key Highlights Regarding Earth’s Atmosphere
- The atmosphere refers to the air enveloping the Earth.
- The thickness of Earth’s atmosphere is roughly 480 km, with 99% of it concentrated within the first 32 km from the planet’s surface.
- As altitude increases, air pressure declines.
- A life-sustaining mixture of gases constitutes the atmosphere.
- Earth’s gravitational force keeps the atmosphere from dissipating into space.
- A primary function of the atmosphere is to block the intrusion of ultraviolet radiation.
- According to NASA, Earth’s atmospheric composition includes:
- Nitrogen: 78%
- Oxygen: 21%
- Argon: 0.93%
- Carbon dioxide: 0.04%
- Small traces of neon, helium, methane, krypton, hydrogen, and water vapo
Composition of Atmosphere

Nitrogen:
Constituting a significant 78.03% of the atmosphere’s total volume, nitrogen was initially identified by Rutherford and bore the moniker “Rutherford” in its early discovery stages. This dominant presence of nitrogen contributes substantially to atmospheric phenomena such as air pressure, wind dynamics, and light refraction. Its properties include being colorless, odorless, tasteless, and retarding rapid combustion. Of paramount importance, nitrogen plays a pivotal role in the synthesis of proteins, facilitating the natural growth of plants, animals, and humans alike. This element finds extensive use in nitrogenous fertilizers, enhancing agricultural productivity.
Oxygen:
Comprising 20.99% of the atmospheric volume, oxygen holds a crucial status as the life-sustaining force essential for respiration, combustion processes, and industrial foundations.
Argon:
At a concentration of 0.94%, argon’s presence in the atmosphere is a result of the radioactive decay of potassium in the Earth’s surface rocks. Slow in its formation rate, argon is categorized as an inert gas, showcasing minimal chemical reactivity.
Carbon Dioxide:
Earning the distinction of being the first gas subjected to scientific examination, carbon dioxide occupies a mere 0.03% of the atmospheric composition. Heavier than many other gases, it predominantly lingers in lower atmospheric layers. Carbon dioxide serves pivotal roles in vegetation growth, contributing to the greenhouse effect, and, when excessive, acting as an asphyxiant gas. Possessing traits of being colorless, odorless, and naturally occurring in various mediums such as air and groundwater, it holds solubility in water. Moreover, its diverse applications encompass functioning as a fire suppressant, aiding in oil recovery, serving as a refrigerant, and facilitating coal bed methane extraction.
Additional Air Components:
The atmospheric milieu encompasses other notable constituents, including neon, helium, methane, krypton, hydrogen, and variable amounts of water vapor.
Role of Earth’s Atmosphere:
- The Earth’s atmosphere consists of gases like oxygen, carbon dioxide, and nitrogen.
- Plants rely on carbon dioxide, while animals and other organisms require oxygen for survival. These essential gases are provided by the atmosphere.
- All life forms need specific temperature ranges and certain frequencies of solar radiation for their biophysical processes.
- The atmosphere selectively absorbs certain frequencies of solar radiation and permits others, thereby regulating solar radiation entry.
- It maintains the Earth’s surface temperature within certain limits, preventing extreme temperature fluctuations between day and night.
- Absence of the atmosphere would lead to stark temperature contrasts on the Earth’s surface.
- The atmosphere, specifically the ozone layer in the stratosphere, blocks harmful ultraviolet radiation from reaching the surface.
- It also safeguards against extraterrestrial objects like meteors, causing them to burn up due to friction as they pass through the mesosphere.
- Weather, a significant phenomenon, shapes various natural and human-driven processes such as plant growth, agriculture, and human settlements.
- Weather results from the interaction of diverse climatic factors.
Structure of Atmosphere
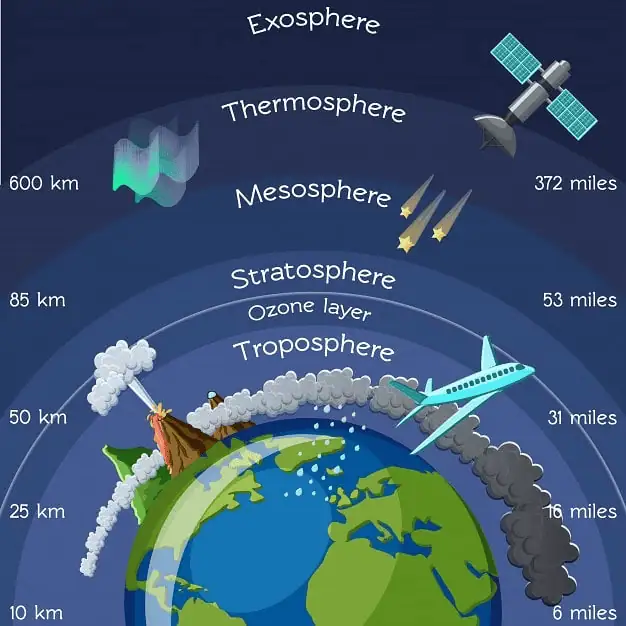
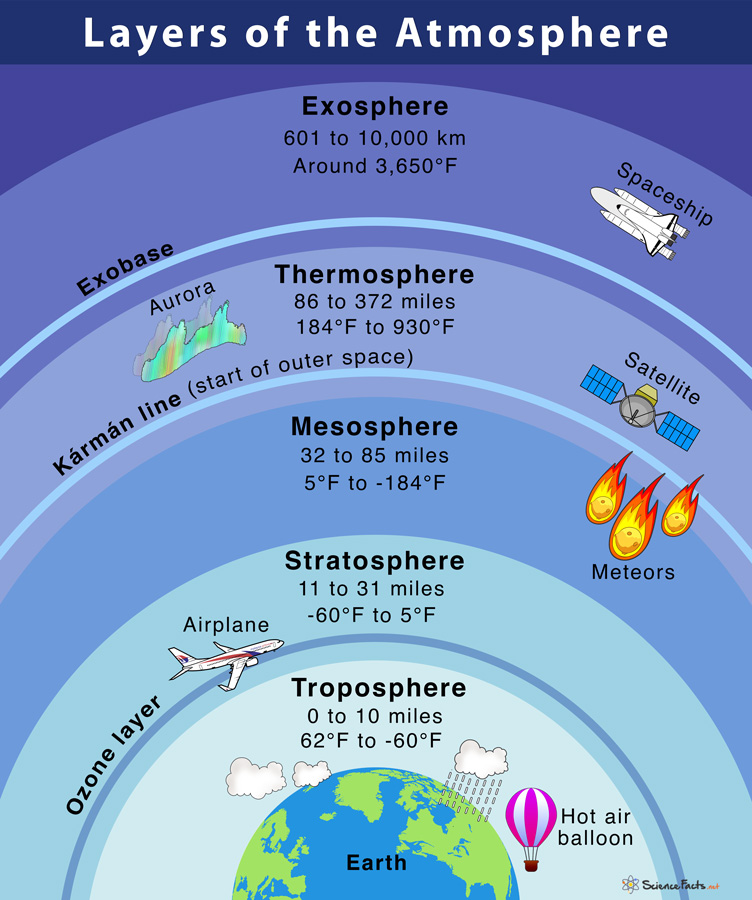
The Earth’s atmosphere is stratified into five distinct layers based on temperature, each serving specific functions:
Troposphere
- The lowest atmospheric layer.
- Extends from the Earth’s surface to about 8 km (at poles) and 18 km (at the equator).
- Weather phenomena occur here due to temperature and pressure variations.\
- Contains water vapor and suspended particles.
- Temperature decreases with altitude, following a normal lapse rate of 1 degree Celsius per 165 meters.
- Tropopause, the boundary between the troposphere and stratosphere, marks its upper limit.
Stratosphere
- Lies above the troposphere.
- Spans from 18 km to about 50 km above the Earth’s surface.
- Low water vapor content.
- Stable horizontal winds and absence of stormy weather benefit aviation.
- Houses the ozone layer, shielding the Earth from harmful UV radiation.
- Separated from the mesosphere by the stratopause.
Mesosphere
- Positioned above the stratosphere.
- Encompasses altitudes from 50 km to 80 km.
- Coldest atmospheric layer, temperature decreasing with altitude.
- Temperature can reach -100 degrees Celsius at its upper boundary.
- Meteors burn up in this layer.
- Mesopause marks the boundary between the mesosphere and thermosphere.
Thermosphere
- Extends from the mesopause, around 80 km, to about 400 km.
- Radio waves are reflected by this layer, aiding communication.
- Temperature begins to rise with altitude.
- Auroras and satellites are present in this layer.
- Lower thermosphere, known as the ionosphere, contains electrically charged particles called ions due to cosmic and solar radiation.
Exosphere
- The outermost layer.
- Reaches from the top of the thermosphere to about 10,000 km.
- Acts as the region where atoms and molecules transition into space.
- It lacks a well-defined upper boundary.
What is green house gas?
A greenhouse gas refers to a gas capable of capturing and re-emitting infrared radiation. These gases absorb the infrared energy, emitted by the Earth’s surface, and subsequently release it back towards the planet’s surface. By doing so, they effectively confine heat within the Earth’s atmosphere, contributing to the warming of the planet.
Major green house gases
Carbon Dioxide:
Carbon dioxide holds significant meteorological importance due to its behavior as a selective radiation absorber. While allowing incoming solar radiation to pass through, it impedes the outward transmission of terrestrial radiation. This dual characteristic prompts the gas to absorb a portion of the Earth’s emitted heat and redirect some of it back towards the planet’s surface. This phenomenon forms the crux of the greenhouse effect, contributing to temperature regulation.
Ozone:
Ozone, albeit present in minute concentrations near the Earth’s surface, wields considerable influence as a greenhouse gas. Its presence, though scarce, is strategically positioned in the stratosphere where it shields the planet from the harmful effects of ultraviolet radiation. This protective function contributes to the overall greenhouse effect by interacting with other greenhouse gases in the atmosphere.
Water Vapour:
Water vapor, a variable gas in the atmosphere, exhibits a vertical gradient in concentration. It diminishes with increasing altitude, and a latitudinal gradient is also apparent—decreasing from equator to poles. The tropics, being warm and moist, can contain up to four percent of air volume as water vapor, fostering a humid environment. Contrastingly, arid and polar regions may see water vapor composing less than one percent of the air.
Water vapor serves a dual purpose in the atmosphere: Firstly, it partakes in the solar energy equation by absorbing specific segments of insolation, thereby regulating temperature. Secondly, it functions as a thermal insulator, retaining the Earth’s radiated heat. This insulating capability is akin to a blanket, ensuring the Earth doesn’t experience extreme cold or heat. Additionally, water vapor influences atmospheric stability, impacting weather patterns and atmospheric behavior.
Methane:
Methane, an imperative greenhouse gas, is formed through the decomposition of organic materials and animal waste. While relatively less abundant compared to other gases, its potent heat-trapping properties make it a critical contributor to the greenhouse effect. Methane’s presence in the atmosphere intensifies radiative forcing, further influencing the Earth’s energy balance and subsequently, its temperature dynamics.
Significance :
- Protection from Harmful Radiations:The atmosphere shields Earth from harmful ultraviolet (UV) radiation, ensuring safety for life by preventing its direct impact. It also maintains a balance of insulation, preventing drastic temperature variations between day and night.
- Weather and Climate Control:The atmosphere is a key player in shaping weather and climate patterns, influencing elements like temperature, rainfall, and wind. These patterns have profound effects on ecosystems, agriculture, and human endeavors.
- Temperature Regulation:Through the greenhouse effect, the atmosphere moderates the planet’s temperature. It captures and retains heat from the sun, preventing excessive heat loss to space. This vital function maintains a suitable temperature for life.
- Water Cycle Management:The atmosphere is a pivotal player in the Earth’s water cycle. By transporting water vapor from oceans to land, it facilitates the process of precipitation, ensuring the distribution of water resources across the planet.
FAQs
Q1: What is the atmosphere?
- The atmosphere is a layer of gases surrounding a celestial body, such as a planet or moon. It plays a crucial role in maintaining life, regulating temperature, and interacting with sunlight.
Q2: What gases are present in Earth’s atmosphere?
- Earth’s atmosphere is primarily composed of nitrogen (78%), oxygen (21%), and traces of other gases like carbon dioxide, water vapor, and noble gases.
Q3: How many layers does Earth’s atmosphere have?
- Earth’s atmosphere is typically divided into five layers: troposphere, stratosphere, mesosphere, thermosphere, and exosphere.
Q4: What is the greenhouse effect?
- The greenhouse effect is the process by which certain gases in the atmosphere, like carbon dioxide and water vapor, trap heat from the sun, keeping the Earth’s surface warm enough to sustain life.
Q5: How does the atmosphere protect us from harmful radiation?
- The atmosphere absorbs and scatters incoming ultraviolet (UV) radiation from the sun, preventing most of it from reaching the Earth’s surface. This protection is crucial for preventing damage to living organisms.
Also Read:- https://www.borthakursiasacademy.com/blog/heat-budget-important-notes-for-apsc/